You were diagnosed with peritoneal mesothelioma. The cancer in your abdomen. Your oncologist mentioned a treatment called HIPEC. Heated chemotherapy. Done during surgery. They said it has changed survival outcomes for patients like you.
This guide explains HIPEC surgery for peritoneal mesothelioma in plain language. You will learn what HIPEC is, why heat is used, how the surgery works, who is a candidate, what your recovery will look like, and what survival outcomes look like for patients who undergo it. The information will help you have a real conversation with your surgical oncologist.

What HIPEC Stands For
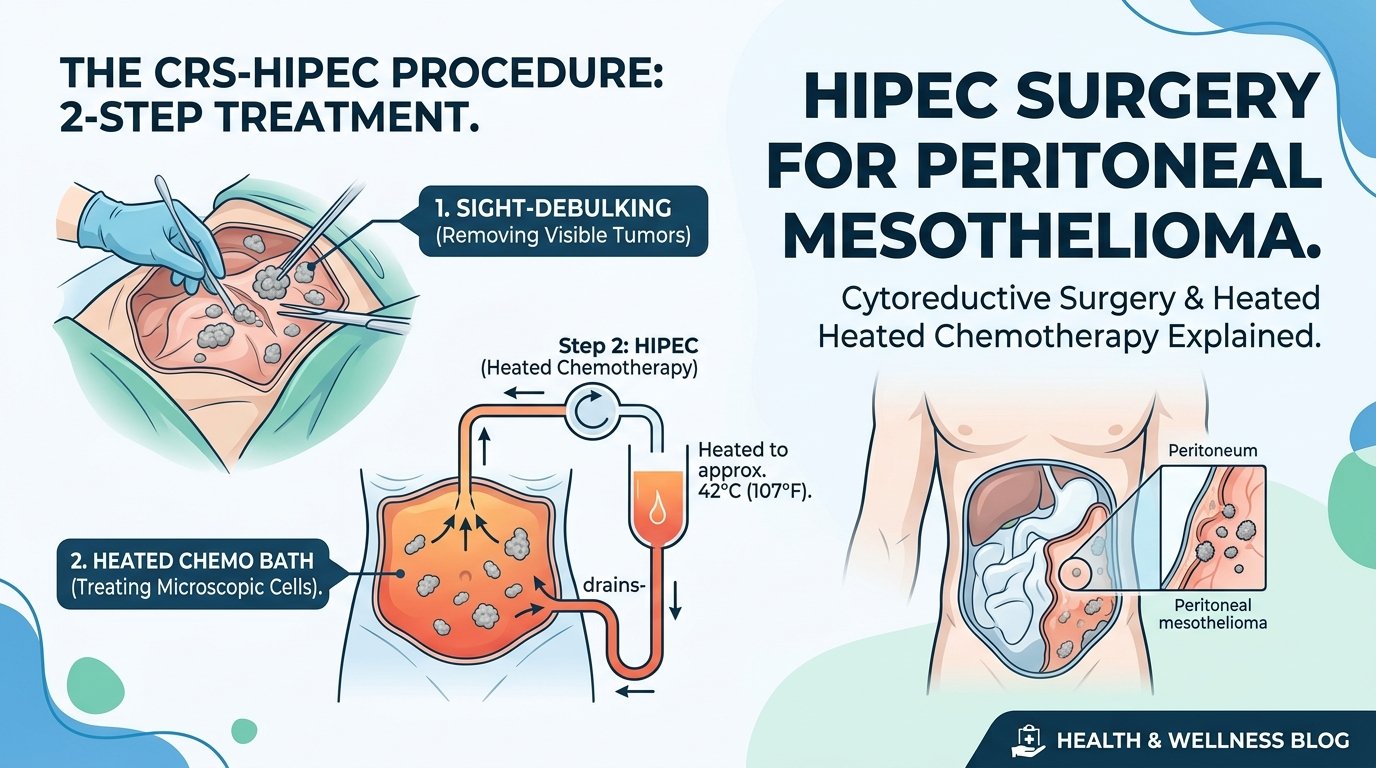
HIPEC is short for hyperthermic intraperitoneal chemotherapy. Hyperthermic means heated. Intraperitoneal means inside the abdominal cavity. Chemotherapy is the cancer-killing drug. So HIPEC is heated cancer-killing drug delivered directly inside the abdomen during surgery, rather than into a vein elsewhere in the body.
The procedure is paired with cytoreductive surgery, also called CRS, which is the surgical removal of all visible mesothelioma in the abdomen. CRS plus HIPEC, performed at the same operation, is the combination that has produced the most dramatic improvements in peritoneal mesothelioma survival over the past two decades. The full name of the operation is therefore CRS plus HIPEC, often spoken simply as HIPEC.
Why Heat Matters
Heat enhances chemotherapy in several ways. It damages cancer cell membranes directly, making the cells more vulnerable. It increases the depth at which the chemotherapy drug penetrates into tissue. It helps overcome certain resistance mechanisms that cancer cells use to survive standard temperature chemotherapy. And it does all of this while the chemotherapy is in direct contact with the surfaces where mesothelioma is most likely to recur.
The chemotherapy is heated to approximately 42 degrees Celsius, about 108 degrees Fahrenheit, and circulated through the abdominal cavity for sixty to ninety minutes. The patient is on the operating table during this time, with the abdomen open or recently closed depending on the technique. Specialised pumps maintain the temperature and circulation. Drainage and inflow catheters are placed to ensure the chemotherapy reaches all surfaces.

The Cytoreductive Surgery Phase
Before HIPEC begins, the surgical oncologist performs cytoreductive surgery. This involves systematically inspecting and removing all visible mesothelioma from the abdominal cavity. Tumour can be growing on the lining of the abdomen, on the surface of the liver, on the spleen, on the omentum, on the bowel surfaces, on the diaphragm, in the pelvis, and elsewhere. The surgeon removes all of it that they can.
This is meticulous work. Hours pass. Each affected surface is dealt with individually. Sometimes organs need to be partially or fully removed when tumour involvement is too extensive to peel away. The omentum, the apron of fatty tissue draping the bowel, is almost always removed because it commonly harbours mesothelioma deposits. The peritoneum lining the abdominal wall is stripped away on affected surfaces.
The completeness of cytoreduction is the strongest predictor of long-term survival. Surgeons grade their result using a scoring system called the completeness of cytoreduction score, with CC-0 meaning no visible disease left and CC-1 meaning only minimal residual disease. Patients with CC-0 outcomes have the best survival statistics. Achieving CC-0 requires both surgical skill and a tumour that is technically resectable.
The HIPEC Phase
Once cytoreduction is complete, the HIPEC begins. The surgical team places inflow and outflow catheters into the abdominal cavity. The chemotherapy, most commonly cisplatin or mitomycin C for peritoneal mesothelioma, is mixed with a carrier solution and warmed to operative temperature. The pump circulates the solution through the abdomen for the prescribed time, typically ninety minutes for mesothelioma protocols.
The patient remains under anaesthesia throughout. The surgical team monitors temperature, fluid balance, kidney function, and other parameters in real time. After the prescribed circulation time, the chemotherapy is drained, the abdomen is rinsed, and the surgery is closed. Drains are placed to manage postoperative fluid collections.
Recovery After CRS Plus HIPEC
The recovery is significant. Hospital stays of fourteen to twenty-one days are typical. The first week is in an intensive care setting. Multiple drains, monitoring lines, nutritional support, and pain management are standard. Bowel function takes longer to return than after most abdominal surgeries because of the combined effects of extensive surgical work and the chemotherapy exposure to the bowel surfaces.
Common complications include prolonged ileus, where the bowel is sluggish to wake up after surgery. Anastomotic leaks if intestinal connections were created during cytoreduction. Wound healing problems. Kidney function changes from the chemotherapy. Infection. Most complications are managed and resolved with time and supportive care, but they extend the recovery and require careful monitoring.
Once home, recovery continues for two to three months before returning to baseline. Energy levels gradually improve. Eating returns to normal slowly. Most patients return to work between three and six months postoperatively, with some delays for those whose work is physically demanding.
Survival Outcomes Have Improved Dramatically
The survival data for CRS plus HIPEC in peritoneal mesothelioma has been transformative. Before HIPEC was widely used, the median survival for peritoneal mesothelioma was less than one year. With CRS plus HIPEC at experienced centres, median survival has reached five to seven years in selected series, with five-year survival rates approaching fifty percent in patients with CC-0 cytoreduction and epithelioid cell type. Some patients are now alive and well a decade or more after their HIPEC operation.
The improvement is so meaningful that peritoneal mesothelioma is now considered one of the most treatable forms of mesothelioma when caught at a stage where CRS plus HIPEC is feasible. This is a notable shift from the prior view that peritoneal mesothelioma was uniformly fatal. The shift is real, and the credit belongs to the surgical and chemotherapy advances embodied in this combined operation.
Who Is a Candidate for HIPEC
Not every peritoneal mesothelioma patient is a HIPEC candidate. The ideal candidate has limited disease that the surgeon can completely remove, epithelioid cell type, no spread outside the abdomen, good performance status, intact heart and kidney function, and the willingness to undergo a major operation with extensive recovery. Older patients can be candidates if their overall fitness is strong.
Patients with sarcomatoid or biphasic cell type have less favourable outcomes and may not benefit as much. Patients with extensive small bowel involvement that would require massive bowel resection are usually not candidates because postoperative bowel function would be inadequate. Patients with metastatic disease outside the abdomen are not candidates because HIPEC only treats the abdominal cavity.
Finding a HIPEC Centre
HIPEC for peritoneal mesothelioma should only be performed at high-volume centres specialising in peritoneal surface malignancies. The list of qualified centres in the United States is in the dozens, not hundreds. The Society of Peritoneal Surface Malignancies maintains member centres. Major academic medical centres including the National Cancer Institute, MD Anderson, Memorial Sloan Kettering, the Washington Cancer Institute, the University of Pittsburgh, and several others have established programmes.
Travelling to a high-volume centre is worth the inconvenience. The difference in outcomes between high-volume and low-volume centres for HIPEC is substantial. Insurance plans generally cover travel and treatment at out-of-network HIPEC centres when the procedure is medically necessary and not available locally. Case managers at the centre can help with the logistics.
A Hopeful Note
Among all the mesothelioma treatment options available today, CRS plus HIPEC for peritoneal mesothelioma represents one of the clearest success stories. The combination of meticulous surgical removal and heated intraperitoneal chemotherapy has changed the prognosis for a disease that once carried a death sentence. The recovery is hard. The risks are real. The benefits, for the right patient at the right centre, are meaningful and durable.
If you have peritoneal mesothelioma, ask whether CRS plus HIPEC is an option for you. If your local team does not perform the procedure, ask for a referral to a high-volume centre. The conversation is worth having early, while the disease is still in the window where surgery can produce a complete cytoreduction.
This article is for educational purposes and does not replace personalised guidance from a surgical oncologist. The Mesothelioma Applied Research Foundation maintains a directory of HIPEC centres.