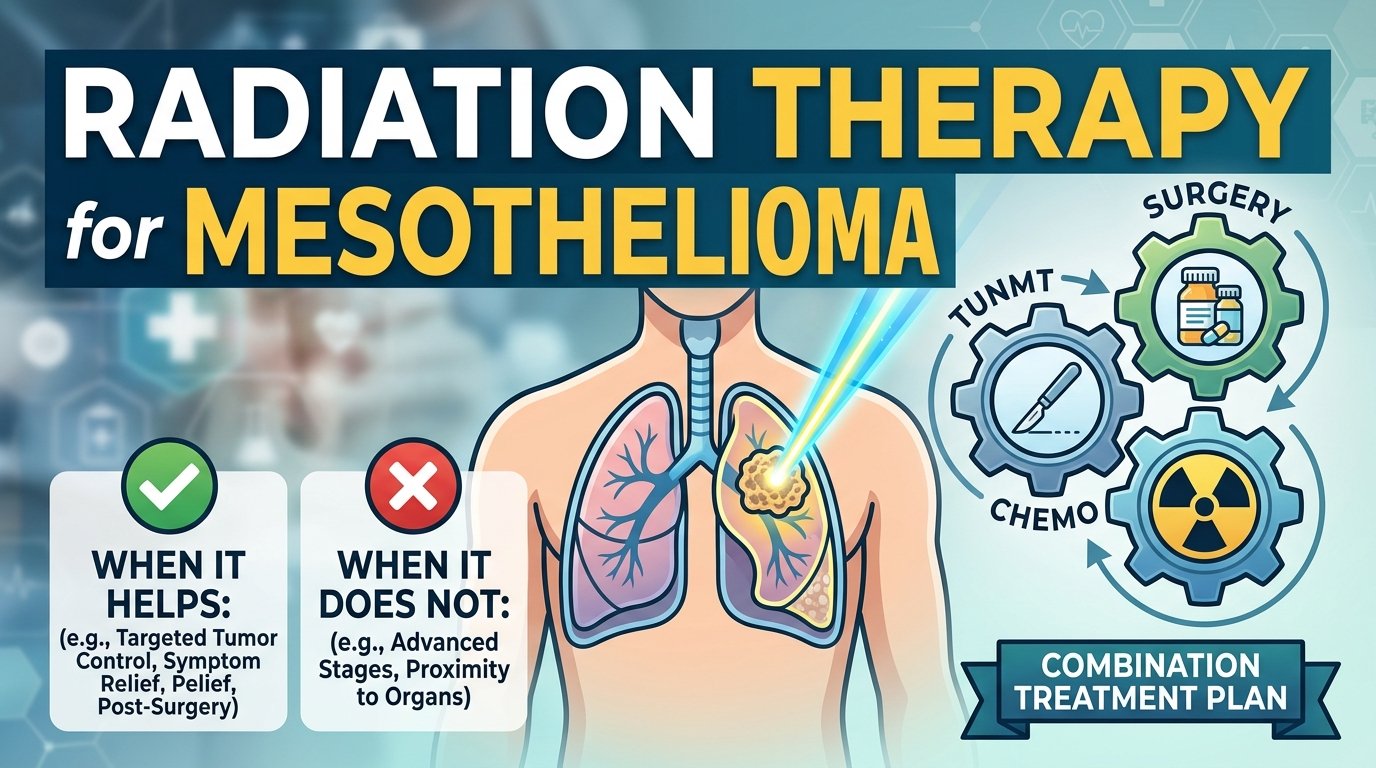
You have been told you need surgery. The doctor wrote down two letters. P slash D. They explained it to you for ten minutes. You nodded. You walked out of the office. You still do not really know what they are going to do to you.
This guide is for you. It explains pleurectomy decortication, often called P/D, in plain language. You will learn what surgeons remove, what they leave behind, why this surgery is sometimes called lung-sparing, what your recovery will look like, what the risks are, and how to know if you are a candidate. No medical jargon for the sake of jargon. Just clear answers.

What P/D Actually Is
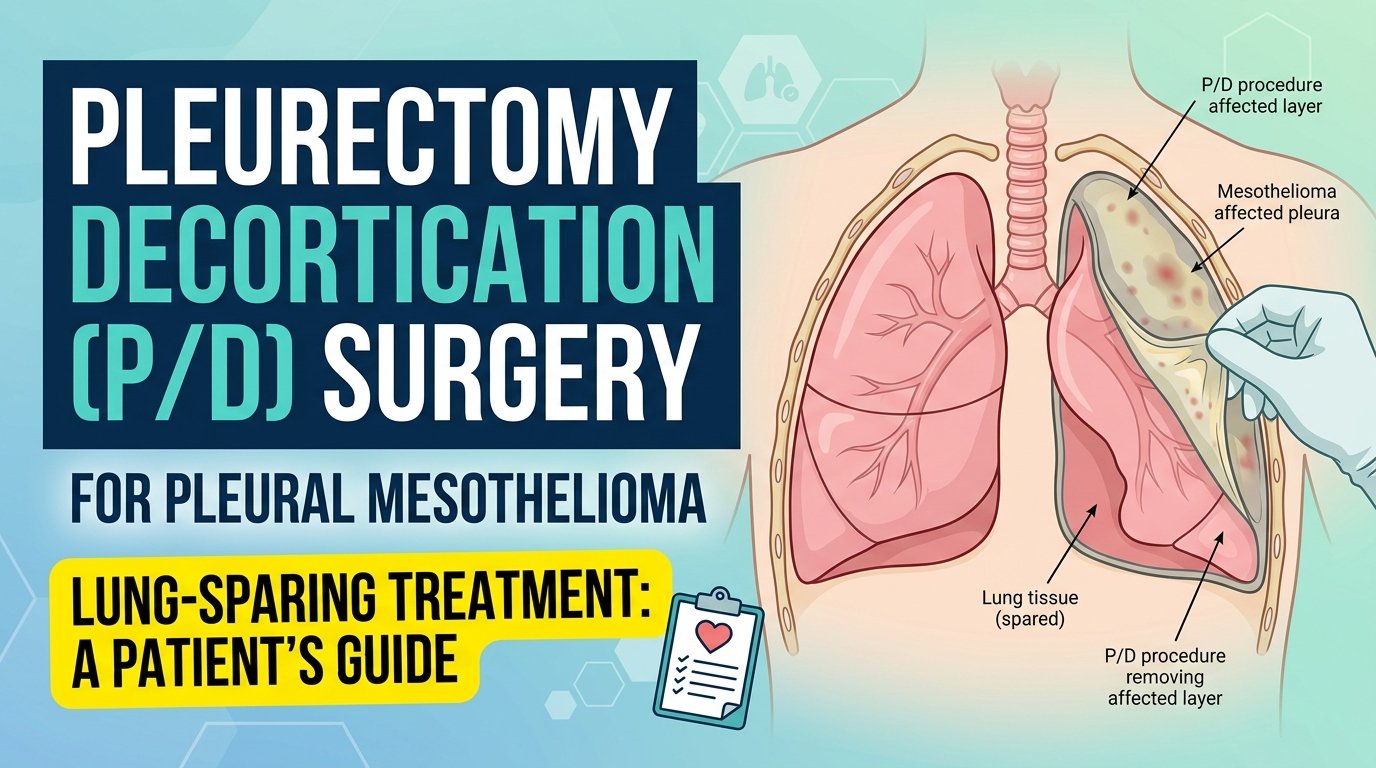
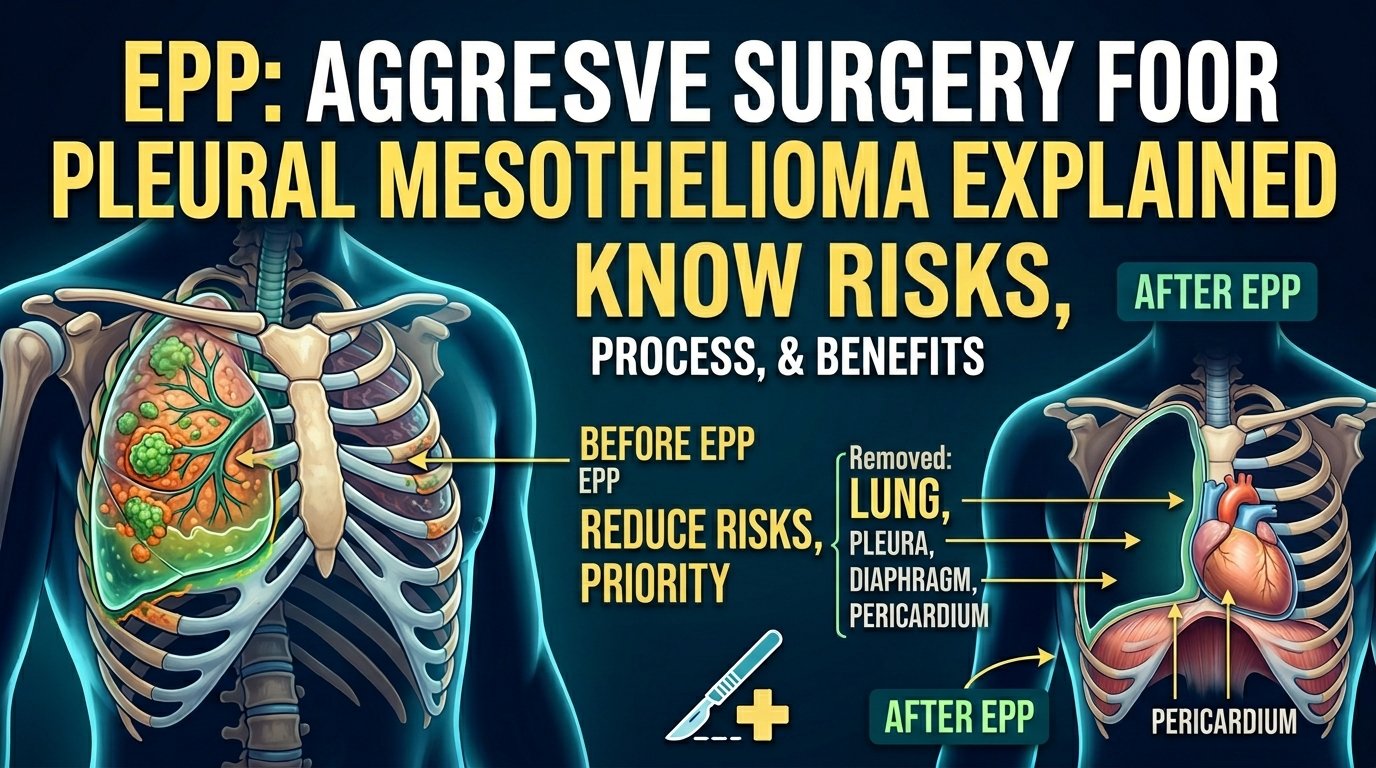
Pleurectomy decortication is a surgery for pleural mesothelioma, the kind that grows on the lining of the lungs. The surgeon makes an incision in your chest, opens the chest wall, and carefully strips away the cancer-affected lining of the lung. They also remove tissue from the diaphragm and the lining of the heart sac when those areas are involved. The lung itself stays in your body. That is what makes P/D different from the more aggressive surgical option, EPP, where the entire lung is removed.
The pleura, the tissue layer that gets removed, is normally only a few millimetres thick. In mesothelioma it thickens with tumour, sometimes to several centimetres. The surgeon’s job is to peel this thickened, cancer-laden lining away from the lung surface and from the chest wall, layer by layer, without damaging the lung underneath. The decortication part of the name refers to this peeling action.
Why Surgeons Choose P/D Over EPP
For decades, the more aggressive operation called extrapleural pneumonectomy, EPP, was the standard mesothelioma surgery. EPP removes the entire lung, the lining, the diaphragm, and the lining of the heart on the affected side. It is a bigger operation with more complications, more deaths during surgery, and a longer recovery.
Studies over the past fifteen years have shown that survival outcomes after P/D are comparable to or better than EPP for most patients. The lower complication rate of P/D, combined with the preserved lung function, has shifted most major mesothelioma treatment centers toward P/D as the first-choice operation. Some surgeons still recommend EPP for specific cases, particularly when the tumour is too extensive to peel away. But P/D has become the default for the majority of operable patients.

Who Is a Candidate
You are likely to be considered for P/D if your mesothelioma is in the early to middle stages, if your tumour is technically resectable based on imaging, if your lung and heart function are strong enough to handle the operation, and if you are otherwise medically fit. Patients with stage I or stage II pleural mesothelioma are the strongest candidates. Some stage III patients qualify if the tumour anatomy allows it.
You will go through a thorough preoperative evaluation that includes a CT scan, a PET scan, sometimes an MRI, pulmonary function tests, an echocardiogram, blood work, and a thoracic surgery consultation. The team is looking for evidence that the tumour can be removed, that you can tolerate the surgery, and that there is no distant spread that would make the operation pointless.
What the Day of Surgery Looks Like
You will arrive at the hospital early, often before sunrise. The pre-op nurses will start an IV, run final blood work, and walk you through what to expect. The anaesthesiologist will meet with you to review the plan. The thoracic surgeon will see you briefly to confirm the operative side and answer any final questions.
The operation itself takes anywhere from four to eight hours. The surgeon makes a long incision along your back and side, between two ribs. They sometimes need to remove a small section of rib to gain access. Once inside the chest, the careful work of peeling the diseased tissue from the lung begins. Surgeons describe this part as both a technical and an artistic process. The pace is deliberate. Every layer must be inspected, separated, and removed without injury to the underlying lung tissue.
Your family will receive periodic updates from the operating room. You will wake up in the recovery area, then move to a thoracic intensive care unit. Chest tubes will drain blood and fluid. A urinary catheter will be in place. Pain control will be managed by a pump that lets you give yourself measured doses on demand.
Recovery in the Hospital
The hospital stay after P/D is typically seven to fourteen days. The first three days are the hardest. You will have several chest tubes, monitoring lines, and significant chest wall pain that the team manages aggressively with medication. By day four or five, the tubes start coming out. By day seven, most patients are walking the hallway with assistance, eating regular food, and beginning to taper off the pain pump.
Pulmonary toileting is constant. Nurses and respiratory therapists will encourage you to use an incentive spirometer hourly, to cough deliberately, and to walk as much as you can. These are not optional. They prevent pneumonia, atelectasis, and the most common postoperative complications. Patients who engage with this work recover faster. Patients who avoid it pay for it later.
Recovery at Home
You will go home with a long incision, residual fatigue, a follow-up schedule with the thoracic surgeon, and often a referral to chemotherapy or radiation as part of the multimodal mesothelioma treatment plan. The first six weeks are about gentle activity, pain management, and graduated return to baseline. Driving usually resumes at three to four weeks. Most patients return to light work between six and twelve weeks. Heavy lifting and physical labour wait longer.
The lung that was peeled re-expands gradually over weeks. Pulmonary function recovers most of its preoperative capacity in most patients, though some loss is expected. Some patients also have nerve pain along the incision site that can persist for months. This is treatable with medications, physical therapy, and time.
Risks You Should Know About
Every surgery carries risks. P/D is a major operation and the risks deserve honest discussion. The surgical mortality rate, defined as death within thirty days of the operation, is in the range of one to four percent at experienced centres. Major complications occur in twenty to forty percent of patients depending on the series. The most common include prolonged air leaks from the lung, pneumonia, atrial fibrillation, blood clots, and bleeding requiring re-operation.
The most important risk-modifying factor is the volume and experience of the surgical team. Mesothelioma surgery should be performed at high-volume centres where surgeons do this operation regularly. The difference in outcomes between high-volume and low-volume centres is substantial and well-documented. If your local oncologist refers you for P/D, ask explicitly how many P/D operations the surgical team performs per year. The answer should be in the dozens, not single digits.
Combining P/D With Other Treatments
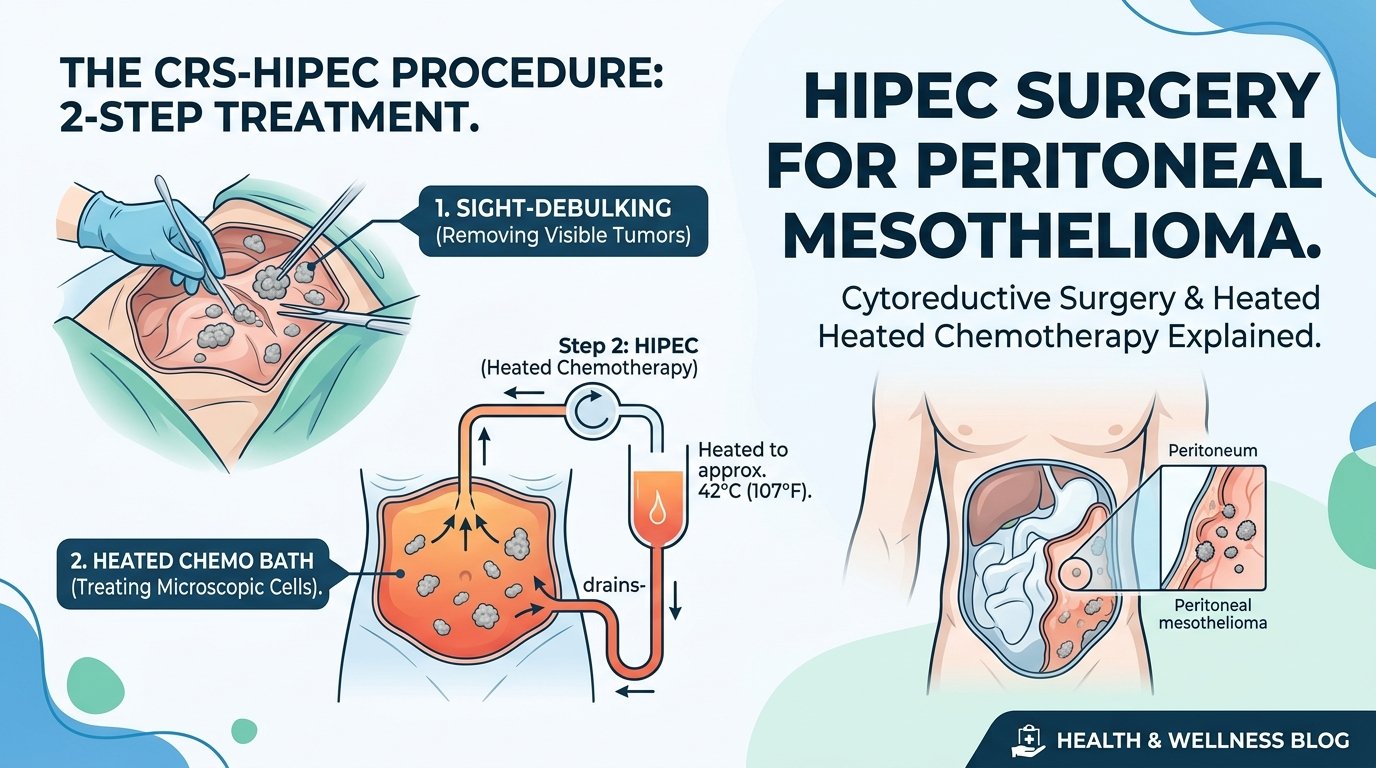
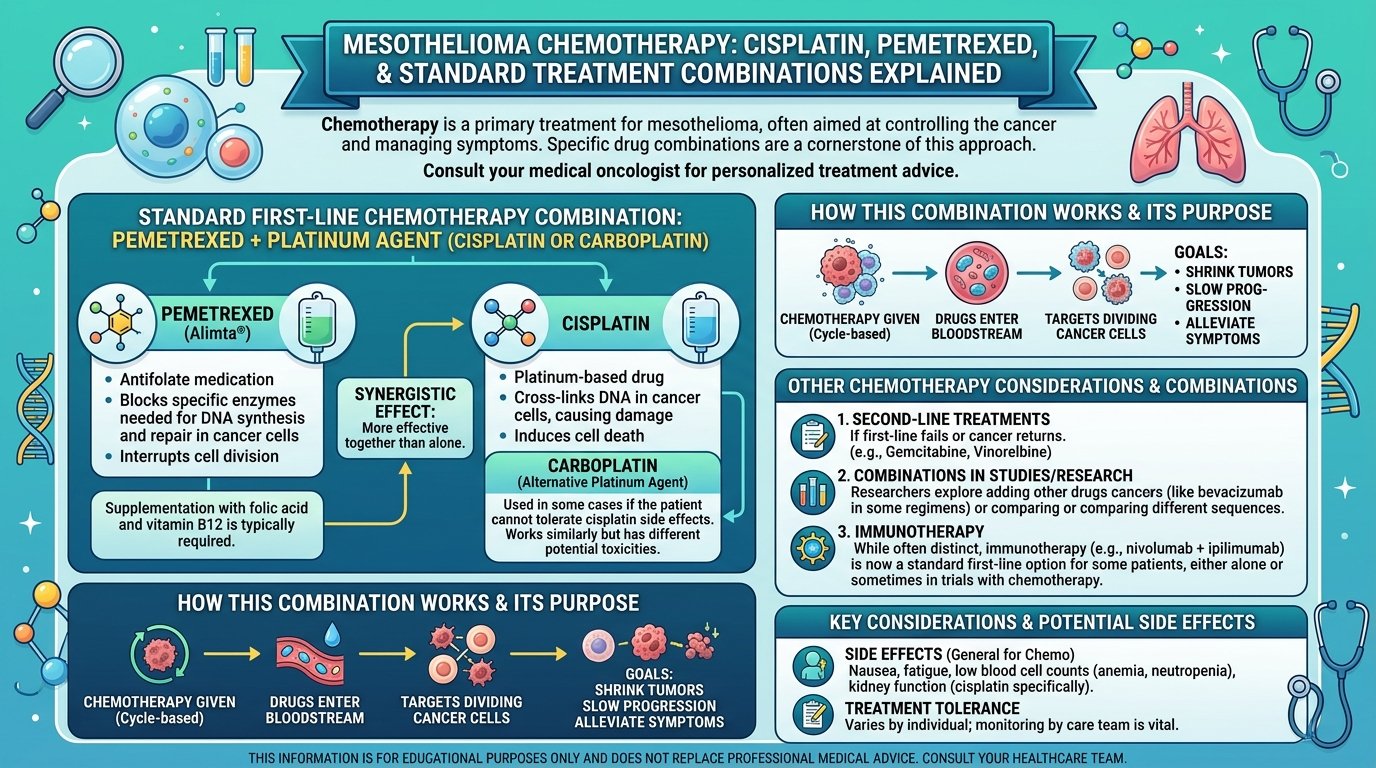
Surgery alone rarely cures pleural mesothelioma. P/D is most effective as part of a multimodal approach that combines surgery with chemotherapy and sometimes radiation. The chemotherapy can be given before surgery to shrink the tumour, after surgery to kill remaining cells, or both. The combinations and timing are individualised based on tumour characteristics, patient fitness, and the experience of the mesothelioma treatment centers managing the case.
Newer immunotherapy combinations are being added to the mix as well, particularly for tumours with specific biomarkers. Clinical trials are testing additional sequences and combinations. Patients considering P/D should ask about clinical trial eligibility as part of the initial workup. Some trials are restricted to patients who are surgery candidates. Missing the window can close off options that may matter.
A Realistic Word About Outcomes
Pleural mesothelioma remains a serious cancer. Even with successful P/D and adjuvant therapy, recurrence is common and median survival from diagnosis is measured in years rather than decades for most patients. That said, P/D has produced meaningful long-term survivors, particularly among patients with epithelioid cell type, lower stage at diagnosis, and good response to chemotherapy.
The decision to undergo P/D is not just a medical one. It is a personal one. The recovery is hard. The risks are real. The benefits are uncertain. The right choice for you depends on your tumour, your fitness, your goals, your support system, and your willingness to undertake an aggressive treatment in pursuit of more time. A thorough conversation with a surgeon experienced in mesothelioma treatment is the starting point. The decision is yours, and it deserves to be made with full information.
This article is for educational purposes and does not replace personalised guidance from a thoracic surgeon or oncologist. For evaluation, contact a high-volume mesothelioma treatment centre. The Mesothelioma Applied Research Foundation maintains a directory of specialty programmes.