You have been feeling off for a while. Pain in your belly. Swelling that will not go away. You have lost weight but your pants are tighter. You feel sick to your stomach. You are tired all the time.
Your doctor ran tests. The results came back. Mesothelioma. But not the kind that affects the lungs. This kind affects your abdomen. It is called peritoneal mesothelioma.
You have never heard of it. You did not even know you could get mesothelioma in your belly. You worked around asbestos decades ago. You thought the danger was to your lungs. You were wrong.
Peritoneal mesothelioma is the second most common type of mesothelioma. It accounts for 10 to 20 percent of all mesothelioma cases. It is caused by swallowing asbestos fibers instead of breathing them in.
This guide is for you. You will learn what peritoneal mesothelioma is, what causes it, what symptoms to watch for, how it is diagnosed, what treatment options are available, what makes HIPEC so effective, what your prognosis looks like, and how to get financial compensation.
No complicated medical language. No confusion. Just clear, honest information to help you fight this disease.
What Is Peritoneal Mesothelioma?
Let us start with the simple explanation.
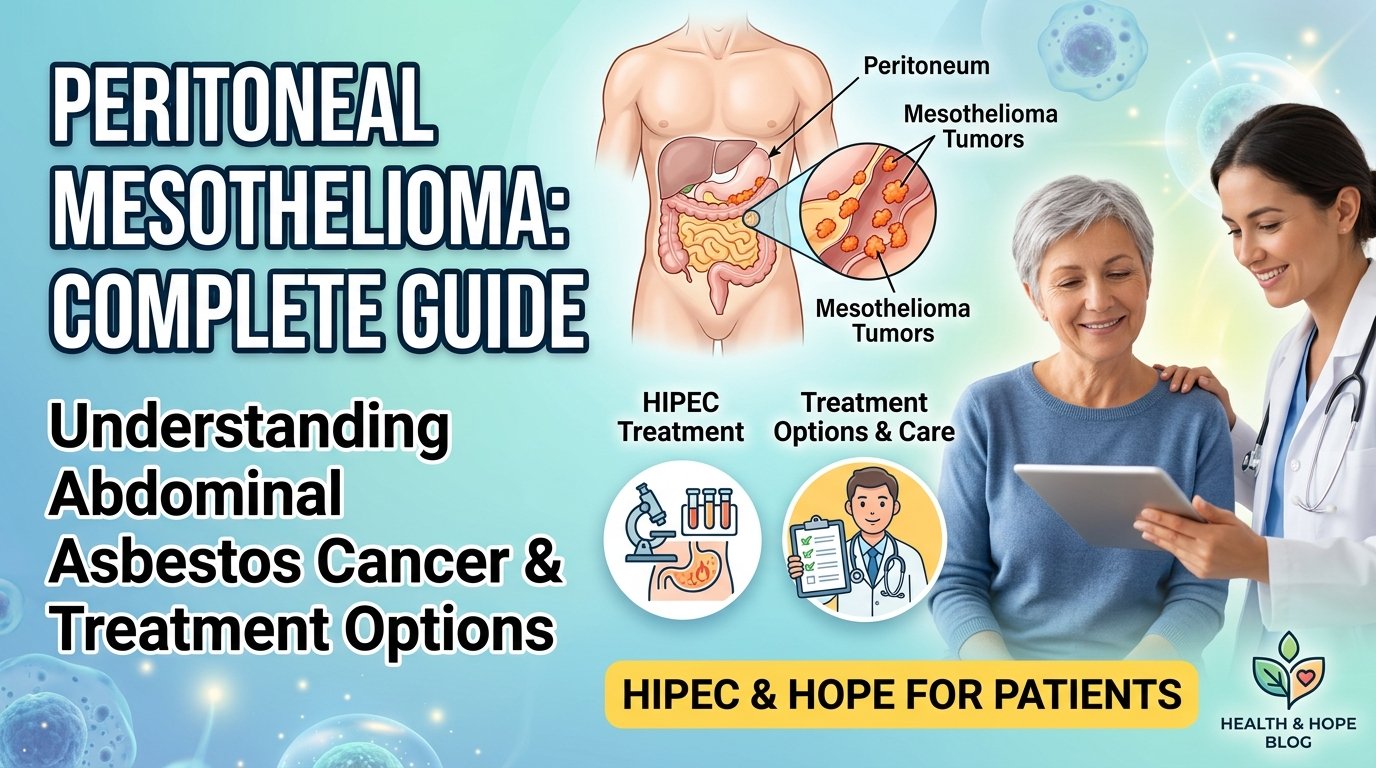
Peritoneal mesothelioma is a cancer that affects the peritoneum. The peritoneum is the thin layer of tissue that lines your abdominal cavity. It covers your stomach, liver, intestines, and other organs. It protects these organs and helps them move smoothly against each other.
When you have peritoneal mesothelioma, tumors grow on this tissue. Fluid builds up in your abdomen. Your belly swells. You feel pain. The cancer can eventually spread to your organs.
The disease is rare. Only about 500 to 1,000 Americans are diagnosed with peritoneal mesothelioma each year. But for those who have it, it is a life-changing diagnosis.
What Causes Peritoneal Mesothelioma?
Peritoneal mesothelioma is caused by asbestos exposure. But here is the difference. Pleural mesothelioma (lung lining) is caused by breathing in asbestos fibers. Peritoneal mesothelioma is caused by swallowing asbestos fibers.
Here is how it happens. Asbestos fibers are tiny. They float in the air. You breathe them in. But your body has ways of clearing them from your lungs. You cough them up. You swallow them. They travel down your throat, through your stomach, and into your digestive system.
Some fibers get stuck in your peritoneum. Your body cannot break them down or get rid of them. They sit there for years, causing inflammation and scarring. Twenty, thirty, forty, or fifty years later, that damage turns into cancer.
Who Is at Risk?
The same jobs that cause pleural mesothelioma also cause peritoneal mesothelioma.
- Shipyard workers
- Construction workers
- Industrial and factory workers
- Plumbers, electricians, and pipefitters
- Navy veterans and other military personnel
- Mechanics
- Family members exposed secondhand through work clothes
If you were exposed to asbestos, you are at risk for both types of mesothelioma.

Peritoneal Mesothelioma Symptoms
The symptoms of peritoneal mesothelioma can be vague. They can look like many other, less serious conditions. That is why it is often misdiagnosed.
Early Symptoms
- Abdominal pain: Pain or discomfort in your belly. It may be a dull ache or sharp pain.
- Abdominal swelling: Your belly may look bigger than usual. This is often caused by fluid buildup (ascites).
- Nausea and vomiting: Feeling sick to your stomach. You may actually throw up.
- Loss of appetite: You are not hungry. Food does not appeal to you.
- Unexplained weight loss: You are losing weight without trying, even though your belly is getting bigger.
- Fatigue: Feeling unusually tired.
Later Symptoms
- Severe abdominal pain: The pain becomes constant and severe.
- Severe abdominal swelling: Your belly becomes very large and uncomfortable.
- Bowel changes: You may have diarrhea or constipation that does not go away.
- Bowel obstruction: The tumor can block your intestines, causing severe pain, vomiting, and inability to have bowel movements. This is a medical emergency.
- Anemia: Low red blood cell count. This can cause fatigue, weakness, and pale skin.
- Fever and night sweats: These are common in many cancers.
- Lumps under your skin: In some cases, tumors can grow under the skin on your belly.
Peritoneal Mesothelioma Symptoms Summary
| Early Symptoms | Later Symptoms |
|---|---|
| Abdominal pain | Severe abdominal pain |
| Abdominal swelling | Severe abdominal swelling |
| Nausea and vomiting | Bowel changes |
| Loss of appetite | Bowel obstruction |
| Unexplained weight loss | Anemia |
| Fatigue | Fever and night sweats |
| Lumps under skin |
How Is Peritoneal Mesothelioma Diagnosed?
If your doctor suspects peritoneal mesothelioma, they will order several tests.
Imaging Scans
- CT scan: This is the most useful scan for peritoneal mesothelioma. It can show tumors in the abdomen, fluid buildup, and whether the cancer has spread.
- MRI: This gives more detailed images of soft tissues, including the peritoneum.
- PET scan: This shows areas of the body that are metabolically active. Cancer cells are very active, so they light up. This helps doctors see if the cancer has spread.
Blood Tests
There is no blood test that can definitively diagnose peritoneal mesothelioma. However, blood tests can look for substances that are often elevated in mesothelioma patients. These tests can help doctors decide if a biopsy is needed.
Biopsy
A biopsy is the only way to know for sure if you have peritoneal mesothelioma. A doctor takes a small sample of tissue from the suspicious area. A pathologist looks at it under a microscope.
The biopsy is often done using a laparoscope. A small camera is inserted through a tiny cut in your abdomen. The doctor can see the peritoneum and take tissue samples.

Peritoneal Mesothelioma Treatment: The HIPEC Breakthrough
The treatment for peritoneal mesothelioma is different from pleural mesothelioma. The standard of care is a combination of surgery and heated chemotherapy called HIPEC.
What Is HIPEC?
HIPEC stands for Heated Intraperitoneal Chemotherapy. It is a two-part treatment done during a single surgery.
Part One: Cytoreductive Surgery (CRS)
The surgeon opens your abdomen. They carefully remove all visible tumors. They may need to remove parts of the peritoneum, parts of the intestines, the gallbladder, the spleen, or other organs if the cancer has spread.
The goal is to leave no visible cancer behind. This is called “complete cytoreduction.” The more cancer the surgeon can remove, the better your outcome.
Part Two: Heated Chemotherapy
After the tumors are removed, heated chemotherapy is pumped directly into your abdomen. The temperature is about 107 to 109 degrees Fahrenheit (42 to 43 degrees Celsius). The heat helps the chemotherapy penetrate the tissues more effectively.
The chemotherapy circulates for about 60 to 90 minutes. It kills any remaining cancer cells that the surgeon could not see or remove.
Then the chemotherapy is drained out. The surgeon closes the incision.
Why HIPEC Works So Well
Peritoneal mesothelioma stays in the abdomen for a long time before spreading. This makes it a good candidate for localized treatment.
HIPEC delivers a very high dose of chemotherapy directly to the cancer. This is much higher than what could be given through an IV. The heat also damages cancer cells directly and helps the chemotherapy work better.
Because the chemotherapy is confined to the abdomen, very little enters the bloodstream. This means fewer side effects than traditional chemotherapy.
Who Is a Candidate for HIPEC?
Not everyone with peritoneal mesothelioma is a candidate for HIPEC. The best candidates have:
- Good overall health (able to tolerate major surgery)
- Cancer that has not spread outside the abdomen
- No other serious health conditions
- Epithelioid or biphasic cell type (sarcomatoid is less responsive)
Your doctor will evaluate you to see if you are a candidate.
HIPEC Outcomes
HIPEC has transformed the treatment of peritoneal mesothelioma. Before HIPEC, life expectancy was less than one year. With HIPEC, many patients live for years.
- Patients who have complete cytoreduction (no visible cancer left) have a median survival of 5 to 7 years or more.
- Patients who have incomplete cytoreduction have shorter survival but still better than without surgery.
- Some patients have lived for 10, 15, or even 20 years after HIPEC.
Other Treatment Options for Peritoneal Mesothelioma
Not everyone is a candidate for HIPEC. There are other options.
Chemotherapy
Systemic chemotherapy (through an IV) can help shrink tumors and relieve symptoms. The standard chemotherapy for peritoneal mesothelioma is the same as for pleural mesothelioma: pemetrexed (Alimta) and cisplatin or carboplatin.
Immunotherapy
Immunotherapy helps your own immune system fight cancer. The drugs nivolumab (Opdivo) and ipilimumab (Yervoy) have been approved for pleural mesothelioma. They are also being tested for peritoneal mesothelioma.
Palliative Care
Palliative care focuses on relieving symptoms and improving quality of life. For peritoneal mesothelioma, this may include draining fluid from the abdomen (paracentesis), pain management, and nutritional support.
Peritoneal Mesothelioma Prognosis and Survival Rates
Peritoneal mesothelioma prognosis has improved dramatically thanks to HIPEC.
Survival Rates with HIPEC
- 1-year survival: 80-90 percent
- 3-year survival: 60-70 percent
- 5-year survival: 50-60 percent
- 10-year survival: 30-40 percent (for patients with complete cytoreduction)
Factors That Affect Prognosis
- Completeness of cytoreduction: This is the most important factor. Patients with no visible cancer left after surgery do much better.
- Cell type: Epithelioid has the best prognosis. Sarcomatoid has the worst.
- Age: Younger patients do better.
- Overall health: Healthier patients do better.
- Treatment center: Patients treated at high-volume centers with experienced surgeons do better.
Peritoneal Mesothelioma vs. Pleural Mesothelioma: Key Differences
| Feature | Peritoneal Mesothelioma | Pleural Mesothelioma |
|---|---|---|
| Location | Abdomen | Lungs |
| Cause | Swallowing asbestos fibers | Breathing asbestos fibers |
| Percentage of cases | 10-20% | 75-80% |
| Primary symptom | Abdominal swelling | Shortness of breath |
| Standard treatment | HIPEC (surgery + heated chemo) | Surgery, chemo, radiation |
| Prognosis with treatment | 5-7 years median | 1-2 years median |
Finding a Peritoneal Mesothelioma Specialist
Not every cancer center has experience with peritoneal mesothelioma or HIPEC. You need a specialist.
Top centers for peritoneal mesothelioma include:
- MD Anderson Cancer Center (Houston, Texas)
- Brigham and Women’s Hospital (Boston, Massachusetts)
- University of Chicago Medicine (Chicago, Illinois)
- University of Texas Southwestern (Dallas, Texas)
- Mayo Clinic (Rochester, Minnesota)
Do not settle for a local surgeon who has never performed HIPEC. Travel if you need to. Your life depends on it.
Your Legal Rights
If you have peritoneal mesothelioma caused by asbestos exposure, you may be entitled to financial compensation.
Asbestos Trust Funds
There are over sixty asbestos trust funds holding more than thirty billion dollars. You can file claims with multiple trust funds.
Lawsuits
You can sue the companies that made the asbestos products that caused your illness. Many patients receive 1millionto2 million or more.
VA Benefits for Veterans
If you are a veteran, you may be eligible for VA disability compensation and free health care.
A mesothelioma lawyer can help you with all of these options. Most lawyers offer free consultations and work on contingency (you pay nothing upfront).
Frequently Asked Questions
Is peritoneal mesothelioma curable?
There is no cure for peritoneal mesothelioma. But HIPEC has helped many patients live for years. Some patients have lived for 10, 15, or even 20 years after treatment.
How painful is HIPEC surgery?
HIPEC is a major surgery. Recovery is hard. You will be in the hospital for 1 to 2 weeks. You will have pain. But the hospital team will manage your pain with medication. Most patients say the recovery is worth it.
What is the recovery time for HIPEC?
Hospital stay: 1 to 2 weeks
Full recovery: 3 to 6 months
Return to light activities: 2 to 3 months
Can peritoneal mesothelioma spread to the lungs?
Yes, in advanced stages. But it usually stays in the abdomen for a long time before spreading. That is why HIPEC works so well.
How do I find a HIPEC surgeon?
Search for “peritoneal mesothelioma specialist” or “HIPEC surgeon.” Top cancer centers have dedicated programs. Call the center and ask to speak with a patient navigator.
What if I am not a candidate for HIPEC?
You still have options. Chemotherapy, immunotherapy, and clinical trials can help. Talk to your doctor.
Hope and Action
Peritoneal mesothelioma is a serious cancer. But it is not the death sentence it used to be. HIPEC has changed everything. Patients who were told they had months to live are now living for years. Some are living for decades.
You need to act. Find a specialist. Go to a top center. Get evaluated for HIPEC. Do not settle for a local doctor who has never treated this disease.
And call a lawyer. The asbestos companies owe you. You can get money to pay for treatment and support your family.
You are not alone. There are doctors, support groups, and other patients ready to help you. Take the first step today.
Disclaimer: This article provides general information about peritoneal mesothelioma, HIPEC treatment, and prognosis. It does not constitute medical advice. Every patient’s situation is different. Always consult with qualified medical professionals about your specific diagnosis and treatment options. If you or a loved one has been diagnosed with peritoneal mesothelioma, speak with a specialist immediately and contact a qualified mesothelioma lawyer to understand your legal rights. HIPEC has changed the outlook for this disease. Do not give up hope.