Your oncologist mentioned a treatment that does not involve drugs or radiation. They called it tumour treating fields. They said it uses electrical fields to slow cancer growth. They said you wear it on your body. You went home unsure whether this is real medicine or science fiction.
It is real medicine. Tumour treating fields, often abbreviated TTFields and sold under the brand name NovoTTF, is FDA-approved for pleural mesothelioma. This guide explains how it works, who it is for, what wearing the device is actually like, and what survival data has been reported.

How TTFields Work
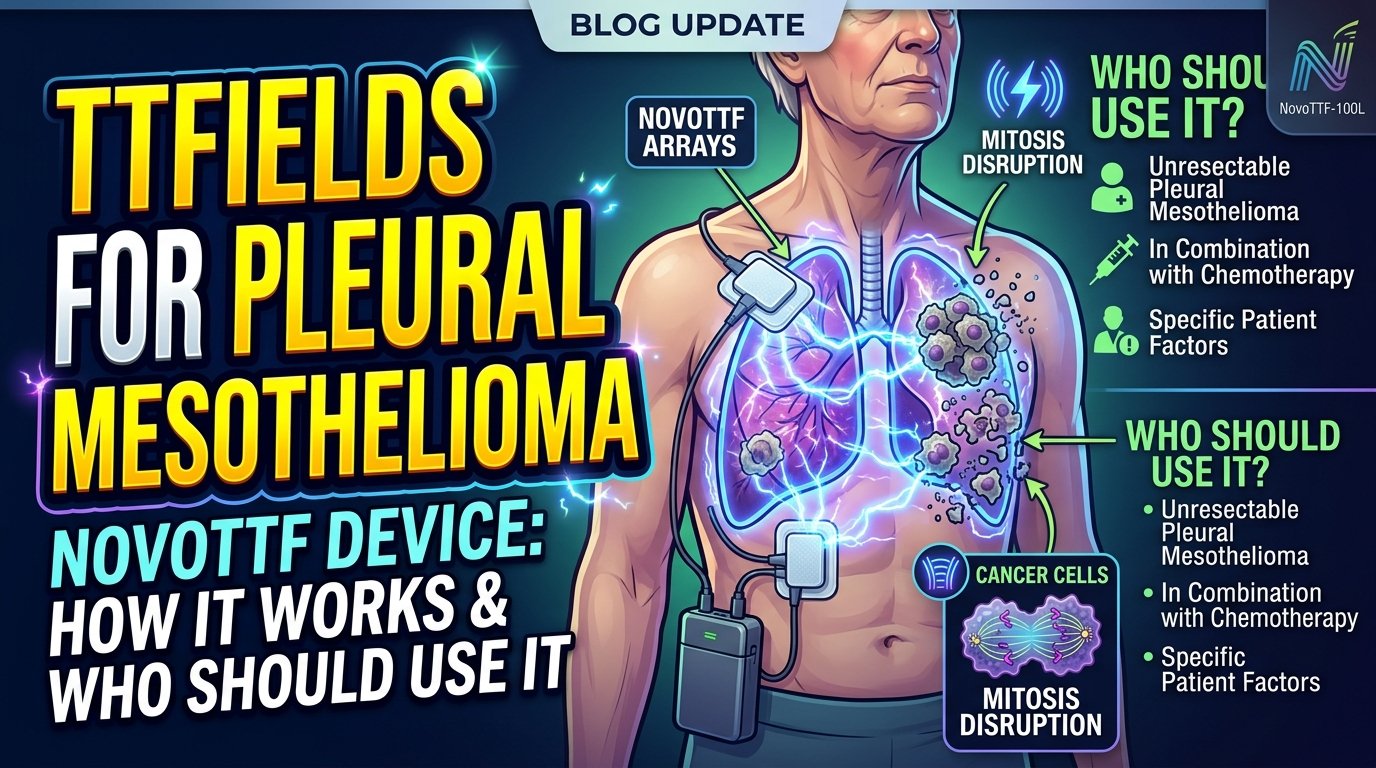
Tumour treating fields use low-intensity, alternating electric fields to disrupt cancer cell division. When a cell divides, certain molecules inside the cell need to align in specific ways to complete the division. The alternating electric fields interfere with this alignment. Cancer cells, which divide much more frequently than healthy cells, are disproportionately affected.
The fields are generated by a portable device the patient wears. Adhesive patches called transducer arrays are placed on the skin of the chest, four arrays per session, and connected by wires to the generator carried in a small bag. The patient wears the device most hours of the day, ideally eighteen hours or more, with brief breaks for showering or skin care.
FDA Approval for Pleural Mesothelioma
The FDA approved TTFields for unresectable pleural mesothelioma in 2019. The approval was based on the STELLAR trial, which combined TTFields with standard chemotherapy and showed median survival improvement compared to historical chemotherapy controls. The approval is for use in combination with first-line chemotherapy in patients who cannot have surgery.
The device is also approved for glioblastoma brain cancer and pancreatic cancer in the United States. The same underlying technology, with different field configurations, is used for each indication. The mesothelioma version is specifically tuned to the body region and tumour location involved.

What Wearing the Device Is Like
The transducer arrays are placed on the chest by a trained technician at the start of treatment. The skin underneath needs to be clean and shaved. Patients shave their chest hair every few days to maintain good contact. The arrays are replaced every two to four days as the adhesive wears out and the skin needs care.
The generator is about the size of a small handbag. It runs on rechargeable batteries that last approximately three to four hours each. Patients carry spare batteries and swap them throughout the day. The device makes a quiet electronic hum but is otherwise unobtrusive. Most patients can continue normal activities with the device on, including light exercise.
The most common side effect is skin irritation under the transducer arrays. Mild redness, itching, and occasionally blistering can develop. Skin care with topical steroids and array repositioning manages most cases. Discontinuation due to skin issues is uncommon when the team is experienced.
Who Is a Candidate
The FDA approval covers patients with unresectable pleural mesothelioma who are starting first-line treatment with chemotherapy. The device is used in combination with chemotherapy, not as a replacement. Patients who are surgery candidates are typically directed to surgery rather than TTFields.
Patients with implanted electronic devices like pacemakers cannot use TTFields because of potential interference. Patients with skin conditions that prevent secure adhesion of the arrays may have difficulty using the device effectively. Patients who cannot tolerate wearing the device for long periods may not benefit because adherence under eighteen hours a day reduces the treatment effect.
Survival Data and Cost
In the STELLAR trial, the median survival with TTFields plus chemotherapy was approximately eighteen months, compared to historical chemotherapy controls of approximately twelve months. The improvement was meaningful, particularly given that there was no increase in chemotherapy-related side effects. Long-term follow-up has continued to show survival benefit, with some patients exceeding three years of survival on the combination.
The device is expensive. The monthly cost approaches twenty thousand dollars before insurance. Coverage varies by plan. Networks behind major commercial insurers and Medicare typically cover the device when used for FDA-approved indications, though prior authorisation is required and the process can be lengthy. The manufacturer, Novocure, has patient support programmes that help with insurance navigation and financial assistance.
Practical Considerations
TTFields is a commitment. Wearing the device eighteen hours a day for many months requires lifestyle adjustment. The visible nature of the device makes hiding the diagnosis from coworkers or social contacts impossible. The skin care, battery management, and array changes become a daily routine.
For patients who can adhere to the protocol, the survival benefit appears real. For patients who cannot or do not want to wear the device consistently, the treatment effect is diminished. The decision should be made with eyes open about the practical demands.
Combining TTFields With Newer Therapies
Trials are now testing TTFields combined with immunotherapy as well as chemotherapy. The mechanisms are independent and may produce additive effects. Early results are encouraging but the formal data is still maturing. Patients on first-line immunotherapy who are interested in adding TTFields should ask their oncologist whether off-label use is appropriate or whether trial enrolment is an option.
The integration of TTFields into broader mesothelioma treatment protocols is still evolving. The next several years will likely see expanded combinations and clearer guidance on when the device adds the most value.
A Closing Note
TTFields represents a fundamentally different approach to cancer treatment than drugs or surgery. The mechanism is novel. The evidence is real. The practical demands are significant. For unresectable pleural mesothelioma patients who can adhere to the wearing schedule, the device offers a meaningful additional tool alongside chemotherapy.
Ask your oncology team whether TTFields is an appropriate addition to your treatment plan. The answer depends on your tumour, your overall treatment strategy, and your willingness to commit to the protocol. The conversation is worth having explicitly rather than assuming the device is or is not relevant to your case.
This article is for educational purposes and does not replace personalised guidance from a medical oncologist.